|
This means that there are 6.02 x 10^23 atoms in a mole. How Many Atom is in a MolesĪ mole is defined as 6.02 x 10^23 atoms. Click here to learn more about the molarity calculators. It assumes you already have solutions of known molarity. 
The calculator will autopopulate other fields where possible. This calculator is designed to output volume measurements when you insert your desired molarity. Then, press the button that says “Convert.” The calculator will then show you the number of moles you have. Molarity Calculator What is Molarity Calculators NOTE: Fill in at least two values to obtain the result of another by clicking the 'calculate' button. First, enter the number of atoms you have. To convert atoms to moles, you need to use a calculator. How do you find moles from molarity If you are given the molarity of any solution and asked to calculate the number of moles, simply multiply the molarity with the total volume of the solution in litres. Number of moles = 2 atoms/6.02 x 10^23 molecules Number of moles = Number of atoms/Atom to mole ratioįor example, if we have 2 atoms of oxygen (O), we can convert this to moles using the above formula. To convert the number of atoms to moles, we use the following formula: A mole is a unit of measurement used to measure a substance’s amount. Atoms are the smallest unit of an element that has the chemical properties of that element. Free online gas law calculator using the van der Waals equation which accepts different input metric units such as temperature in celsius, fahrenheit, kelvin pressure in pascals, bars, atmospheres volume in both metric and imperial units cubed. 
The molecular weight can be obtained from the molecular formula, data tables, or the label on the bottle containing the chemical of interest.What is The Difference Between Atoms To Moles?Ītoms and moles are two different ways of measuring the amount of a substance. A versatile Van der Waals calculator with which you can calculate the pressure, volume, quantity (moles) or temperature of a gas, given the other three. 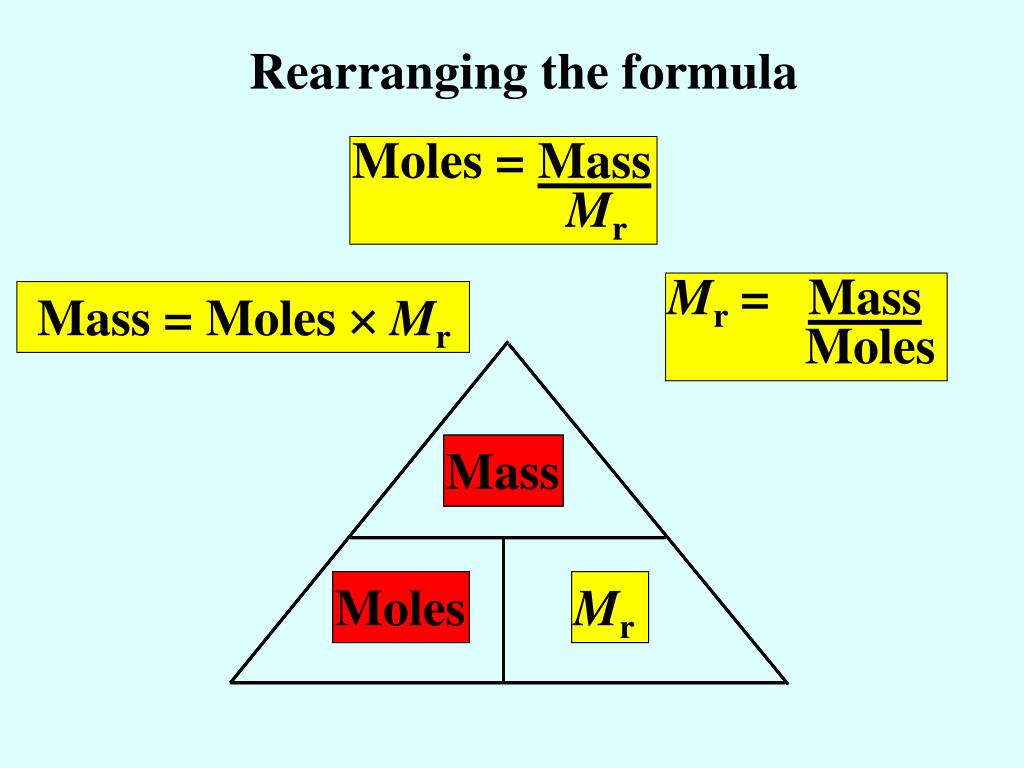
First convert the mass of oxygen to moles of oxygen: 1 mol O2. Molecular weight is also referred to as formula weight and, in fact, many scientists prefer to use the latter. EXAMPLE: Calculate the volume of 0.250 g of oxygen at 25oC and 742 mm Hg pressure. Note that V is the final or total volume of solution after the solute has been added to the solvent. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
